
Former Obama chief of staff and Mayor of Chicago, Rahm Emanuel famously said in 2008: “You never want a serious crisis to go to waste. …it’s an opportunity to do things that you think you could not before.”
Well, outside of war, America has had few national crises worse than this COVID-19 pandemic. And unfortunately, few have provided a greater opportunity for those who crave power to vastly expand it.
The pandemic has now resulted in the deaths of over 500,000 Americans and economically impacted nearly every family and small business. As bad as this has been, there is another enormous issue for the American people: The failure of those we rely on and entrust to prevent such catastrophes — or at least mitigate their damage. And perhaps even more alarming is the possibility that this failure is not due to incompetence — but intent.
I believe this saga to be one of the greatest medical scandals in American history. Perhaps like the New York state nursing home scandal, this saga will eventually receive the national media attention it deserves but until then it remains largely ignored.
After a year of dealing with this pandemic, it is time to look back and evaluate the performance of the federal healthcare bureaucracies, the national media and the entire medical community that we’ve historically and faithfully entrusted with our lives.
This is the amazing saga of what came to be mockingly known as “Trump’s drug,” a.k.a., hydroxychloroquine (HCQ), a 65-year-old, safe medication primarily used to treat lupus and rheumatoid arthritis along with its original use as a prophylaxis for malaria.
As Yale professor of epidemiology, Harvey A. Risch says, “This misbegotten episode regarding hydroxychloroquine will be studied by sociologists of medicine as a classic example of how extra-scientific factors overrode clear-cut medical evidence.”
For many of us, it’s shocking to discover that a year of dealing with this deadly virus and with all of our medical / health care bureaucracies —[Centers for Disease Control and Prevention (CDC), National Institutes of Health (NIH), National Institute of Allergy and Infectious Diseases (NIAID), Food and Drug Administration (FDA), Health Resources and Services Administration (HRSA) and many others]— that the official federal guidance on treating someone who’s elderly, at-risk and has just tested positive for COVID-19 is to stay at home, rest, stay hydrated and take Tylenol. Whoop-de-do.
As Dr. Peter McCullough, vice chair of Internal Medicine at Baylor University Medical Center, said in testimony at a recent Senate hearing, “(NIH) Treatment Guidelines direct doctors to let even high-risk COVID-19 patients sicken at home for two weeks or more and when finally gasping and choking for air, place them in hospital isolation.”
“(NIH) Treatment Guidelines direct doctors to let even high-risk COVID-19 patients sicken at home for two weeks or more and when finally gasping and choking for air, place them in hospital isolation.”
HISTORY OF HCQ
Chloroquine was developed by (a German company) Bayer in the 1930s and to combat malaria. It was widely used by American troops after it fell into Allied hands during WWII. Later on it was modified to produce hydroxychloroquine (HCQ), a superior malaria-fighter that is safer and less toxic than chloroquine. It was approved by the FDA in 1955. Virtually every American soldier who served in Korea or Vietnam or where malaria was prevalent was given chloroquine or hydroxychloroquine.
In the 1950s, researchers found that HCQ had potent anti-inflammatory and anti-viral properties. It helps suppress cytokine signals in immune responses making it effective for autoimmune conditions like lupus and rheumatoid arthritis for which it is still prescribed today.
The drug is now believed to have therapeutic potential for HIV, Hepatitis C, melanoma, pancreatic cancer, Hashimoto’s thyroiditis, ovarian cancer, breast cancer, lung cancer, cystic fibrosis and others.
There are few drugs in existence that have proven to be as safe and effective as HCQ. For seven decades, millions of people have taken billions of doses without incident. There have been zero reported fatal arrhythmias with the medication since its approval and just three fatal overdoses.
HCQ was again studied as recently (2014) on its effects in children of women with lupus who have used it long term. No problems were discovered and the authors concluded, “Hydroxychloroquine should be continued during pregnancy and breastfeeding.”
 Even today on its website, the CDC says “Hydroxychloroquine can be prescribed to adults and children of all ages. It can also be safely taken by pregnant women and nursing mothers.” You might be shocked about that if you’ve only listened to the media about the drug.
Even today on its website, the CDC says “Hydroxychloroquine can be prescribed to adults and children of all ages. It can also be safely taken by pregnant women and nursing mothers.” You might be shocked about that if you’ve only listened to the media about the drug.
PANDEMIC BACKGROUND
We know that the virus began in Wuhan, China likely in the fall of 2019. The World Health Organization (WHO) said recently it was unlikely that the virus spread from the Wuhan lab; that animal to human contact was more plausible. Maybe time will tell us its exact origins but the epicenter was certainly Wuhan.
At the same time the Chinese Communist Party was doing all it could to deceive and deflect blame, many dedicated Chinese physicians were working diligently to contain the virus and communicate with colleagues in the West.
The CDC says “Hydroxychloroquine can be prescribed to adults and children of all ages. It can also be safely taken by pregnant women and nursing mothers.”
They found early on that none of the 178 patients initially admitted into Wuhan’s hospital for COVID-19 had lupus. This was a surprise to them as lupus is an immune disease which should have made its patients especially vulnerable to COVID. Since these lupus patients had been routinely receiving HCQ, the doctors thought it was worth a closer look.
As the virus was spreading rapidly, Chinese doctors initiated a study of 10 nearby hospitals in January 2020. They concluded in a published study that HCQ was effective against COVID-19-related pneumonia.
Shortly thereafter, on March 18th, the Chinese physicians published the results of another study in the journal, Nature, showing that HCQ was effective in blocking the virus in a test tube making it potently “antiviral.” This was all very encouraging. The authors also verified the drug decreased inflammation (which was already known since the body’s inflammatory processes can go haywire with illnesses like lupus and rheumatoid arthritis).
By March it was also recognized that COVID-19 causes what is known as an often fatal, “cytokine storm” in many at-risk patients. Inflammatory cytokines are immune system proteins. COVID can cause the immune system to explosively erupt, producing so many cytokines that this “cytokine storm” attacks the body’s own cells. In COVID patients the tissue that is most vulnerable to cellular destruction is in the lungs.
So the lab scientists were encouraged since HCQ (1) had antiviral properties, (2) was an anti-inflammatory compound and most importantly, (3) it was safe.
This was about as far as lab scientists could take the examination of HCQ against COVID-19. From here on it would require studies involving actual humans.
* * *
In March 2020, one of the world’s foremost infectious disease specialists, physician and microbiologist, Dr. Didier Raoult, immediately began to study HCQ against COVID-19. He directs the l’Institut hospitalo-universitaire (IHU) in Marseilles, France.
No one in Europe has been professionally cited more than Dr. Raoult, who has identified 468 novel species of bacteria. His publishing resume contains more than 2,000 papers.
Dr. Raoult is a brilliant, fearless, colorful eccentric researcher. He’s a 68-year-old motorcyclist who still treats patients. And he’s been a major player in the HCQ / COVID-19 controversy.
Dr. Raoult has had a decades-long familiarity with HCQ. He spent part of his childhood taking chloroquine in French Senegal (Dakar) where his father was a military physician. He considered the findings of the Chinese physicians exciting enough to warrant further study using humans.
Most have heard the old medical adage that “antibiotics kill bacteria, not viruses.” While true for most antibiotics, it is surprisingly not the case for the antibiotic, azithromycin. Dr. Raoult’s group previously had demonstrated that azithromycin did indeed have antiviral effectiveness.
One of Dr. Raoult’s specialties has been the “repurposing” of inexpensive generic drugs for new uses. If a safe, existing drug can be found to fight a new illness, only the drug’s efficacy is an issue as physicians would have years of knowledge and comfort about its safety — its long term effects on major organs or how it interacts with other drugs.
The Raoult researchers decided to find out whether a “drug cocktail” of HCQ combined with azithromycin had efficacy against COVID-19. The first small study involved just 36 COVID patients. 14 received HCQ alone and 6 got HCQ plus azithromycin for 10 days. 16 were in the “control group” who received neither (patients from another nearby hospital who were either not offered the drugs or refused them outright).
By the sixth day, 70% of the HCQ group no longer tested positive for COVID compared to just 12.5% in the control group. Of those who got both drugs, all six or 100% were COVID-free by the fifth day.
Further analyses by the Raoult group found that these medications had to be administered very soon after symptoms began. This is not unusual. Anti-flu drugs, for example, must also be taken early to stop the virus before its takes hold.
What Dr. Raoult and his team showed early in the pandemic was that we now had a possible, yet not proven, but certainly possible protocol to dramatically reduce the number of hospitalizations (remember this was the original goal of “flattening the curve”) and ultimately reduce the number of deaths from this deadly disease.
It was an admittedly very small study but the researchers believed the results were positive enough to quickly publish it for frontline physicians to try themselves. Given the proven safety record of the medications, the risk of trying this HCQ cocktail to at-risk patients was extremely low and given that the number of fatalities, especially in Europe at this time was beginning to explode, there was little to lose.
It was now mid-March and President Trump had begun daily news conferences with a newly formed, coronavirus task force.
But it was at the White House news conference on March 19th where President Trump made what he believed was an optimistic and encouraging remark about new evidence that appeared to show HCQ could be a promising discovery in the battle against COVID-19.
The president announced that early studies, including that by Dr. Raoult’s team in France, revealed that HCQ could be a “game-changer” in the fight against COVID-19. But when Dr. Anthony Fauci, head of the National Institute of Allergy and Infectious Diseases (NIAID), was asked if he thought the drug was promising against COVID, he said, “The answer is no.”
Dr. Fauci’s comment was interesting given the fact that he had no idea if HCQ could be a “game-changer” at the time. He said, “It was not done in a controlled clinical trial. So you really can’t make any definitive statement about it.” But his response, “The answer is no” is about as definitive as you can get.
It’s possible the president was set up to mention HCQ and thus forever taint the drug with the media but more likely he was naive enough not to consider the political ramifications when he made the hopeful “game-changer” remark. Little did he know at the time that it would forever link him with the drug and unite those who were already politically and ideologically against the president with those economically against HCQ.
HCQ was now officially “politicized.” It became “Trump’s Drug” and any objectivity in analyzing its efficacy against Coronavirus would forever be severely compromised.
On March 21st President Trump doubled-down when he tweeted: “HYDROXYCHLOROQUINE & AZITHROMYCIN, taken together, have a real chance to be one of the biggest game-changers in the history of medicine…”
The following week, the president was proactively taking action to ensure that the U.S. would have a large stockpile of HCQ by making a deal with India, which produces most of the world’s supply.
At this time “only” about 200 Americans had died from COVID-19 but even this early in the fight, the battle lines had been drawn over this mostly unknown medication. HCQ was now officially “politicized.” It became “Trump’s Drug” and any objectivity in analyzing its efficacy against Coronavirus would forever be severely compromised.
HYDROXYCHLOROQUINE = “TRUMP’S DRUG”
By late March, while red states (with Republican governors) began to quickly seek out the drug, blue states were showing an aversion to “Trump’s drug.” The media almost celebrated the tragedy of an Arizona couple who ignorantly ingested an aquarium cleaning product that contained chloroquine resulting in the man’s death. It fit their narrative of the idiotic Trump supporter blindly following their leader’s marching orders.
CNN and others in the anti-Trump media not surprisingly, jettisoned all objectivity regarding HCQ and launched into a nonstop campaign attacking HCQ’s safety. What they did not report was that those who were denied early treatments and ended up on ventilators in hospitals had an 80% chance of dying.
When Michigan state Rep. Karen Whitsett, a Democrat, thanked President Trump and gave HCQ credit for saving her life —that her physician was able to acquire even though Gov. Gretchen Whitmer had issued an executive order forbidding its use in the state for COVID— she was excoriated by the media and her fellow Democrats. Her Democratic colleagues formally censured her for promoting the drug and “endangering the health, safety and welfare of her constituents…”
However, Dr. Raoult’s early researched did pave the way for the FDA to issue an emergency use authorization (EUA) on March 28th permitting hospitals to administer HCQ to COVID-19 patients outside of any clinical trials.
DR. ROBIN ARMSTRONG
In April, other frontline physicians decided to share their successes in the hope what worked for them could save lives elsewhere.

Dr. Robin Armstrong, is an internal medicine physician and medical director at a nursing home in Texas City (near Galveston). He reported that in early April his staff was confronted with a sudden outbreak of COVID-19 among staff and 38 elderly patients. He said these were the “sickest of the sick.” Virtually every patient had “coronary heart disease and chronic medical issues.”
Dr. Armstrong said, ““We treated these patients with a five day regimen. On day one we gave them 400 mg of HCQ, twice a day. On day two through five we gave them 200 mg twice a day. We gave them a normal Z-Pak (azithromycin), 500 mg on day one and 250 mg daily on days two through five and we gave them 220 mg of zinc daily.”
He said they were closely monitored for their oxygen saturation and any cardiac arrhythmias.
Dr. Armstrong added, “We had no side effects from the HCQ in any way… This was a success story and you would have thought that the media would have celebrated it as such but certainly they did not. … It’s amazing to me the negative responses we’ve gotten from the national press.” Of the 38 patients, two “very frail” patients died later on from what he said were unknown causes.
“Physicians are taking this themselves when they get COVID-19, they’re prescribing it for their families when they get COVID-19 but they will not prescribe it for their patients because they’re afraid they’re going to get in trouble.”
“I believe the FDA and the medical boards across the country have been responsible for the deaths of many people. .. Because they have been putting restrictions on this drug, hydroxychloroquine. I believe thousands of Americans, tens of thousands of Americans have died unnecessarily,” Dr. Armstrong lamented.

This headline from NPR was typical of the national media, “COVID-19 Patients Given Unproven Drug In Texas Nursing Home In ‘Disconcerting’ Move.”
Dr. Armstrong said perhaps the most disturbing reaction to his nursing home’s success was the negative reaction by states’ medical boards. He said he has physician friends who are petrified to prescribe HCQ to their patients.
“Physicians are afraid that they’re going to lose their licenses. Physicians are taking this themselves when they get COVID-19, they’re prescribing it for their families when they get COVID-19 but they will not prescribe it for their patients because they’re afraid they’re going to get in trouble,” Dr. Armstrong bemoaned.
CLINICAL STUDIES
Recall that on March 19th only 50 Americans were dying daily of COVID-19. Four days later the toll would be 100 per day. Ten days after that 1,000 were succumbing to COVID-19 every day in the U.S. This was at a time when places in Europe were experiencing an even higher number of COVID cases and deaths.
It is within this context that the ethics of the “gold standard” study that Dr. Fauci was (still is) advocating —a randomized, double-blind, placebo-controlled trial (RCT)— is highly questionable according to Dr. Raoult. He argued that the only ethical study method during a deadly pandemic is the “observational” study.
With milder diseases or illnesses, RCTs often give solid results without placing the placebo-receiving patients at serious risk. But COVID-19 is deadly, often within a few weeks. There is no moral justification for the use of a lottery to determine which patients might be designated for death. This is medical Russian Roulette.
Clinical trials/studies are literally human experiments. In the case of COVID-19, the “observational” study, in which patients and/or their families knowingly agree to take the drug which they know up front could, potentially, save their lives. The outcomes of these patients are then compared to other COVID-19 infected patients with similar demographics and comorbidities but either didn’t have access to the drug or declined to take it.
Objectives of Covid-19 Studies
There are four main areas where treating COVID-19 infected patients with HCQ (or any drug) could have efficacy: (1) Pre-exposure prophylaxis, (2) post-exposure prophylaxis or early treatment, (3) symptomatic with the disease firmly rooted and (4) advanced stages of the disease.
Dr. Raoult’s researchers focused on (2) early treatment. There are few, if any, diseases in which starting treatment early is not beneficial. His findings and now (as of February 2021) the results of dozens of other subsequent observational studies (and “meta-analyses”) have been enormously positive.
In any clinical trial whether it be an RCT or an observational study, researchers must control or adjust for “confounding factors” or physical differences like obesity, cardiac issues, diabetes, immunodeficiencies and others.
It’s a fallacy that observational studies don’t have control groups like RCTs do. These are often groups with similar demographics and confounding factors but in different settings or locations. Observational studies must simply equalize the groups after-the-fact or retrospectively. This is exactly what Dr. Raoult did. He didn’t withhold treatment from anyone, but found similar patients elsewhere who, for one reason or another, didn’t receive the drugs.
Observational Studies
The observational study is the backbone of internal medicine. Most of the history of medical science is based on the observational study. An analysis of the guidelines for treating 41 various infectious diseases as identified by the Infectious Diseases Society of America (IDSA) and officially recommended between 1994 and 2010 found that more than half relied on — observational studies including 37% based the guidelines on “expert opinion only.”
The observational study is the backbone of internal medicine. Most of the history of medical science is based on the observational study.
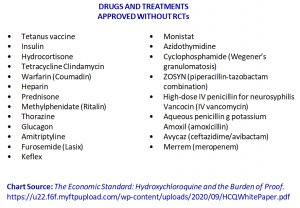
In 2011, the Journal of the American Medical Association (JAMA) published a scientific study that sought to find out what percentage of existing American College of Cardiology (ACC) and American Heart Association (AHA) guidelines or treatment protocols had been established by RCTs. The findings are truly astonishing. Just “… 8.5% of recommendations in ACC/AHA guidelines … were classified as level of evidence A (supported by evidence from multiple RCTs)” or fewer than one out of eleven.
Two of the most commonly used medications and their initial approvals were not based on RCTs. Where would diabetics be today without insulin? Or how about doing without a tetanus vaccination? Both of these have been used for decades but neither was approved based on the “gold standard” RCTs to prove their efficacy.
Some scientists have studied studies. Two studies compared the results of both RCTs and observational studies and found there is little difference between them. One analysis was done in 2000 and another in 2014 by an epidemiologist in New Zealand. Both came to the same conclusion that there was little differences in results of the two primary methods.
Many researchers say that the “gold standard” study or RCT should perhaps be considered more of a “silver standard” or maybe even a “bronze standard.” Princeton University’s Angus Deaton, says RCTs can be weak foundations for inferring efficacy. Dr. Deaton adds RCTs have value but only pertain to “averages” not toward specific patients who are only truly known medically by their personal physicians.
Even former CDC director (appointed by President Obama) Thomas Friedan, in a 2017 article in the New England Journal of Medicine (NEJM), wrote that “limitations” (cost, logistics and lengthy time to conduct) “affect the use of RCTs for urgent health issues, such as infectious disease outbreaks, for which public health decisions must be made quickly on the basis of limited and often imperfect available data. RCTs are also limited in their ability to assess the individualized effect of treatment…”
This underscores the tension and differences between physicians who have frightened COVID patients in front of them desperately seeking treatment protocols and the “removed” clinical researcher who demands ideal methodology.
Dr. Raoult agrees that in a perfect world, an RCT using a placebo would provide the best answers but it is unethical to intentionally give a sugar pill to an elderly, comorbid patient with deadly COVID-19 when the next randomly selected patient gets the drug that could possibly save his or her life. The face-to-face doctor, must deal with the human being in front of him or her — now.
Put another way, if you had 2 groups of COVID patients, and you actually said to the patients, “You have a disease that might kill you. We want to see if drug “ABC” works on reducing your breathing difficulty and might even save your life. Would you like to take this drug that has been safely given to millions of people over the last 65 years or would you prefer a sugar pill and take your chances?”
How many do you think would take the drug?
Or how about this: If you get COVID the CDC recommends you take acetaminophen. No problem taking Tylenol, right? But if you were told that you were in a study in which drug XYZ (acetaminophen) was being tested for its efficacy against COVID and found out that XYZ (acetaminophen) was responsible for “nearly half of the cases of acute liver failure in the United States and remains the leading cause for liver transplantation” according to the NIH, would you still take XYZ (acetaminophen)? Maybe not.
* * *
The worldwide demand for HCQ was exploding as increasing numbers of frontline physicians reported impressive successes. An international poll of 6,200 doctors in 30 countries was taken in late March and revealed that HCQ was the number one drug of choice in dealing with COVID-19. Of 15 possible choices, 37% chose HCQ as “the most effective therapy.”
The New York Times reported that in the United States, pharmacists were getting “alarmed” that doctors were perhaps stockpiling HCQ for themselves and families.
Even in the U.S. a survey of 1,271 physicians in all 50 states found nearly two-thirds of doctors would treat their family members with HCQ for COVID-19. 30% said they would also prescribe it to a loved one prophylactically. Just 11% had an aversion to the drug.
The New York Times reported that in the United States, pharmacists were getting “alarmed” that doctors were perhaps stockpiling HCQ for themselves and families.
One day after the Times’ article, India, the world’s largest producer of HCQ announced it would no longer export the drug. It claimed that it was holding back on exports until additional clinical trials were conducted. Another one of the world’s largest producers, Hungary announced essentially the same thing.
Not much of a surprise really when a country may possess the ability to make a possible cheap but effective in dealing with the worst and deadliest pandemic in a century.
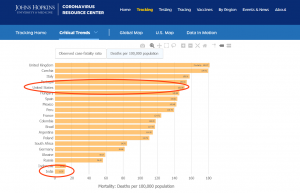 [It’s worthwhile to note that, as Dr. Peter McCullough told a Senate Homeland Security and Governmental Affairs Committee hearing in November, that because HCQ is given to COVID patients as soon as they show symptoms in India, that (much poorer) country has a death rate per 100,000 of just one-eighth or 13% of that in the United States.]
[It’s worthwhile to note that, as Dr. Peter McCullough told a Senate Homeland Security and Governmental Affairs Committee hearing in November, that because HCQ is given to COVID patients as soon as they show symptoms in India, that (much poorer) country has a death rate per 100,000 of just one-eighth or 13% of that in the United States.]
Some (mostly “blue” or those with Democrat governors) states were even beginning to ban the ability of physicians to prescribe HCQ. For example, North Carolina’s Board of Pharmacy took action to deny these prescriptions in late March. Its statement said that too many doctors were prescribing HCQ for themselves and their loved ones. But if they tried to save their patients they either could not get the drug or risked the wrath of their state’s licensing board.
“To my knowledge, neither governors nor boards of pharmacy have ever outlawed any legal drug – not even opioids like Oxycontin that cause about 30,000 deaths a year.”
Dr. Lee Merritt, a fellowship-trained orthopedic spine surgeon declared, “To my knowledge, neither governors nor boards of pharmacy have ever outlawed any legal drug – not even opioids like Oxycontin that cause about 30,000 deaths a year.”
ZELENKO STUDY
As the media continued to try to destroy Dr. Raoult’s reputation for promoting now “Trump’s drug,” his American counterpart, Dr. Vladimir Zelenko, a board-certified family practitioner in New York state near West Point, quickly became a new target.
Dr. Zelenko’s crime was his discovery that adding zinc to Dr. Raoult’s HCQ “cocktail” was proving very effective to his COVID patients. In early April, he reported he had successfully treated about 200 mostly elderly patients with this HCQ cocktail plus zinc. By the end of the month the number was up to 400 elderly patients treated with HCQ, azithromycin and zinc with just two had succumbing to the illness, a rate far lower than what New York state was experiencing everywhere else with its elderly residents.
Many months later, other studies backed up Dr. Zelenko’s inclusion of zinc in the treatment protocol. A study published in November said zinc deficiency in COVID-19 patients resulted in “more complications” and “prolonged hospital stays.”
Not surprisingly, the “cancel culture” shifted into high gear. Dr. Zelenko’s videos were quickly censored on YouTube for “disinformation.” The New York Times also applied the “conspiracy theorist” to Dr. Zelenko along with trashing his professional reputation by saying he was a compromised Trump supporter, making his research an unscientific political invention. He was disparagingly referred to as a “Right-Wing Star.”
What these media outlets chose to ignore was that the 1,448 of the 1,450 high-risk, elderly COVID-19 patients who Dr. Zelenko treated with HCQ — didn’t die. This statistically important find was considered a minor detail and deemed irrelevant by the national media. He continued to treat hundreds of COVID-19 patients with a 99.9% success rate.
Additionally, his outpatient drug regimen costs only $12.00 — 200mg of HCQ twice a day for 5 days, 500mg of azithromycin once per day for 5 days and 220mg of zinc sulfate for 5 days. Like Dr. Raoult, he said the key is early treatment. It keeps people out of hospitals, off ventilators and truly flattens the curve so those same hospitals are not overrun.
Dr. Zelenko, with the assistance of two German researchers, produced a retrospective study based on his detailed findings and was published last fall. Of the 144 patients in the study, one (0.7%) (who had a history of cancer and received just one dose of the medications before being hospitalized) died. In the control group of 377 patients from the same community who were not treated with these medications, 13 (3.4%) died.
* * *
The following five studies likely had the most (negative) impact in the debate about the effectiveness of HCQ. They were all published within 90 days of each other.
VA STUDY
The first “scientific” knockdown punch against HCQ came on April 21st when the American Veteran’s Administration (VA) released a study that claimed HCQ was not only ineffective for hospitalized patients, it was dangerous to many. The VA study was funded by the National Institutes of Health (NIH) and University of Virginia.
It was a “retrospective” analysis of 368 patients in all VA medical centers: 158 received neither HCQ nor azithromycin, 97 got HCQ alone and 113 received HCQ plus azithromycin.
“In this study, we found no evidence that use of hydroxychloroquine, either with or without azithromycin, reduced the risk of mechanical ventilation in patients hospitalized with Covid-19,” the researchers said.
The results of the non-peer reviewed study were heavily trumpeted in the media and embraced by Washington bureaucrats.
When such studies are widely published, most readers assume the study is at least, reasonably objective; that each of the groups analyzed are comparable. But for this VA study, even the authors wrote that was not the case.
The authors said they tried to adjust for “confounding” factors but said they may have missed important ones. They wrote, “Hydroxychloroquine, with or without azithromycin, was more likely to be prescribed to patients with more severe disease, as assessed by baseline ventilatory status and metabolic and hematologic parameters.” HCQ use and timing was left to the attending physicians so it was no surprise that the sickest patients received the drug likely as a last chance effort, after everything else had failed, to save their lives.
Many were not put on HCQ until after they were intubated, meaning they were very ill. And for the 90 patients given HCQ earlier in the illness, just 7.8% were later intubated versus 14.2% of the 177 patients in the control group who were eventually put on ventilators. This 45% reduction in intubation was not mentioned or clarified when specifically pointed out to the authors.
CNN’s Anderson Cooper seemed thrilled to report that this cheap, safe, widely available drug was now deemed ineffective and dangerous. Under the banner, “…No Benefits, Higher Death Rate,” Cooper said you risk losing your life if you take HCQ for COVID-19.
Now it was Dr. Raoult’s turn to pounce. He said the authors failed to mention that the HCQ-receiving patients were much closer to death than the “standard” treatment group as evidenced by their low white blood cell count, typical of late COVID patients. There were twice as many with low white cell count in the HCQ group as in the control group.
Robert Wilke, Secretary of Veteran Affairs admitted as much. He said the study was conducted on a “small number of veterans, sadly those of whom were in the last stages of life. …We know that the drug has been working on middle aged and younger veterans … In stopping the progression of the disease.” (My emphasis)
Dr. Raoult also stated a number of the patients were given HCQ and azithromycin after they were put on ventilators, many who had severe organ failure due to a cytokine storm. This was clearly way too late. Additionally, 30% of the “standard” treatment group had at times been given azithromycin.
Dr. Raoult said the media reaction to HCQ was astounding. They were now saying a drug that had been around for over 65 years with billions of prescriptions written was suddenly one of the most dangerous drugs in the world.
At the time, investigative journalist Sharyl Attkisson was reporting that the media were claiming there were two primary reasons why HCQ should not be used to treat COVID-19 patients: (1) “We need to keep supplies for people with rheumatoid arthritis and lupus, who are dependent upon it,” then immediately adding, (2) “besides, the drug is a dangerous killer.” Which is it? Both cannot be true.
After the VA Study was made public, Neil Cavuto warned his viewers on his Fox News TV program: “If you are in a risky population here, and you are taking this (HCQ) as a preventative treatment to ward off the virus, or worst-case scenario you are dealing with the virus, and you are in this vulnerable population, it will kill you. I cannot stress enough, this will kill you.”
Moments later Cavuto interviewed Dr. Janette Nesheiwat who told him, “Neil, so it’s important to understand, first and foremost, this is an FDA-approved drug. It is not new, it’s been around for many years, and he is taking it as a prophylactic, preventative measure, which I think is very smart to do.” Cavuto was incredulous.
“LANCET” STUDY
On May 22nd, one of the most astonishing medical studies ever published appeared in The Lancet, the second most prestigious medical journal in the world. At the time it was considered the likely final nail in the coffin for HCQ.
This “Lancet” study was actually authored by Dr. Mandeep Mehra, a professor of medicine at Harvard University. It claimed to be an enormous retrospective, peer-reviewed, observational study of 96,000 patients who had been treated in 671 hospitals and 30 countries. The study supposedly captured data between December 20, 2019 and April 14, 2020.
It should have raised red flags immediately. For a study of this magnitude to be compiled, interpreted, peer-reviewed and published in just five weeks was logistically implausible.
The authors concluded that giving HCQ to hospitalized patients resulted in a higher death rate (30%) and more ventricular arrhythmias in the patients who survived. This news came on the heels of the VA study which had already heavily discouraged use of HCQ.

It became headline news in every major newspaper in the world. One of the primary portrayals was that the American president had foolishly promoted a drug that was killing people.
The Guardian, one of Europe’s most important newspapers, said the study tainted the work and previously stellar reputation of French physician and researcher, Dr. Didier Raoult as well. He was now deemed, at best, “controversial.”
The Washington Post seemed almost gleeful in announcing, “Antimalarial drug touted by President Trump is linked to increased risk of death in coronavirus patients, study says.”
“Malaria Drug Taken by Trump Is Tied to Increased Risk of Heart Problems and Death in New Study” said The New York Times’ headline.
It’s difficult to overstate the worldwide shockwaves produced by the publication of this study. Immediately a number of international RCTs involving HCQ were suspended or shut down due to (1) ethical concerns about giving very sick patients a purportedly dangerous drug and (2) the supply of willing volunteers quickly drying up.
The Hycovid and Discovery studies, two “gold standard” RCTs in France were stopped mid-stream though both were showing early positive effects from the use of HCQ. The Hycovid study reported a decrease in fatalities of 46% when it was stopped.
One of the largest studies being conducted by the World Health Organization immediately suspended its clinical trial of HCQ. Other studies were interrupted in Belgium and Italy.
The National Institutes of Health halted its funded RCT on the effectiveness of HCQ in hospitalized patients. While it did say the drug was safe, it concluded it provided no benefit so it stopped the study mid-stream.
It appeared the “Lancet” study the one the FDA was waiting for and it responded promptly. It ignored the controversy surrounding the it and on June 15th it revoked its Emergency Use Authorization (EUA) that it had previously granted for HCQ for hospitalized patients in late March.
Following the revocation of its EUA, on July 1st the FDA published a safety review of the adverse effects for the 85 days that the drug was “legally” used in hospitals. Even with thousands of doses distributed over those 11 weeks to some of the sickest patients, the FDA found only 5 adverse side effects.
One wonders then what the ulterior motive was for revoking the ability of physicians to prescribe a drug no more dangerous than Advil.
This action by the FDA immediately derailed other ongoing studies. Dr. Jon Giles, a rheumatologist a Columbia University, could no longer recruit patients for his HCQ study. Dr. Giles expressed frustration as he had prescribed HCQ safely for many years.
______________________________________________________________________
The FDA has a history of dubious drug rejections and approvals. For example it approved Purdue Pharma’s now infamous painkilling drug, Oxycontin, in 1995. It did so even though Purdue’s clinical trials had not been successful. The FDA apparently overlooked those and took Purdue’s word that it was safer than most competing painkillers since it was believed to reduce the abuse liability. David Kessler, the head of the FDA at the time, said he had no involvement at all in Oxycontin’s approval process. He said the overseer was Dr. Curtis Wright who left the FDA soon after the drug was approved to accept a position at — Purdue Pharma.
______________________________________________________________________
Just four days after the “Lancet” study was published, Dr. Raoult in France launched an unprecedented attack accusing the authors of falsifying the data to reach a predetermined conclusion.
It was startling since it was from one of the world’s foremost researchers against a professor at Harvard and published in one of the world’s most prestigious medical journals.
Dr. Raoult said there were too many oddities in the study for it to be plausible. For example, 66% of the doses administered were much higher than that recommended by the FDA in its EUA. Of course any patient outcomes would be poor if their physicians were overdosing them.
There were only two possible fallouts to Dr. Raoult’s accusation. Either he was correct and this would be one of the greatest scandals in medical history —or— his reputation would be permanently destroyed.
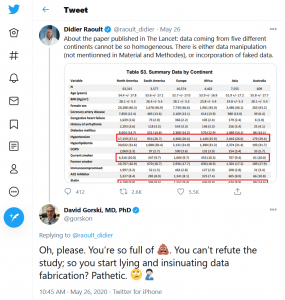
Dr. Raoult was attacked for his comments almost immediately. David Gorski, a professor of medicine and editor of the website, sciencebasedmedicine.com said on Twitter that Dr. Raoult was “pathetic” and a liar for attacking the “Lancet” study.
But very shortly after Dr. Raoult publicly put out his criticisms of the “Lancet” study, scientists from around the world also jumped on board.
The first red flag was the credibility of Surgisphere, the company that provided the data to the authors. Some of the hospitals it cited didn’t even exist.
Surgisphere’s data was also used by most of the same authors in a separate study published shortly afterward in the New England Journal of Medicine (NEJM). This study said that HCQ produced heart problems in hospitalized patients with COVID-19.
On June 4th, over 100 researchers signed an open letter to Dr. Mehra and the other authors along with Richard Horton, the editor of The Lancet delineating their “integrity concerns” about the study. They asked the authors to immediately share their raw data so they could be examined.
The authors declined. Surgisphere, is run by Dr. Sapan Desai, one of the lead authors in both the “Lancet” study and that published in the NEJM. (He also happens to be the brother-in-law of another author, Dr. Amit Patel.) Surgisphere had fewer than a dozen employees at the time the data were being compiled. One of the company’s primary employees was a porn actress, another a science fiction writer. You can’t make this up.
When sharing of the raw data of the study was denied, The Lancet broke a promise it made just two months before when, according to Allysia Finley of the Wall Street Journal, it promised to share all data as it related to COVID-19.
Once the authors were cornered, they requested their study be withdrawn. In doing so they listed the companies which provided “personal fees” to them in the recent past. Included were pharmaceutical or medical equipment giants Abbott, Medtronic, Bayer, FineHeart, Astra Zeneca, Novartis, Amgen and others.
On June 4th, NEJM retracted its study.
The following day The Lancet also issued a formal retraction for the Mehra study.
But the damage had been done.
From (London’s) The Guardian: “The World Health Organization and a number of national governments have changed their Covid-19 policies and treatments on the basis of flawed data from a little-known US healthcare analytics company.”
Given the enormous repercussions of the publication of this study effectively shutting down sincere, scientific attempts to find out about the efficacy of HCQ in hospitalized COVID-19 patients, the question becomes — as it has been asked repeatedly during this pandemic — was there intent to deceive or was it gross incompetence?
Keep in mind that this study was “peer reviewed” before it was published. The purpose of peer review is to ensure the integrity of scientific research. The primary medical journals are submitted a considerable amount of papers/studies whose authors would like to see published. Generally, the editors of these journals ask 3 or more experts to examine the manuscript and actively request clarifications, revisions or reject it outright. In this case, the process unquestionably failed. Thus far, the editors have refused to provide the names of the peer reviewers.
Following the publication of the fraudulent study, the editors of the two publications scrambled to save their reputations. The Lancet’s Horton called the study a “fabrication” and “monumental fraud.”
Dr. Eric Rubin, the editor-in-chief of NEJM, said, “I’m an infectious disease doctor, I treat COVID-19 patients. I’ve been in the hospital recently treating patients, and we have no idea what to do.”
Desperation? Maybe. But how many times have you heard over the last year about “listening to the science” and “trusting the experts?”
There may have been other more subtle reasons for hurriedly publishing these “studies.” Remember this is “Trump’s drug” we’re talking about. The Lancet’s editor, Richard Horton, may have had another motivation other than to advance the cause of “science.”
In past years a study of this magnitude would likely have taken many months to verify prior to publication. Could this nearly unprecedented speed to publication been due to political influences entering the scientific sphere? Could a visceral bent against Trump and by proxy, “Trump’s drug,” have been a factor?
Well, Horton was publicly anti-Trump as evidenced by an editorial in his publication on May 16th, just 6 days before the “Surgisphere” study was published.
Horton’s publication wrote that because of Trump’s “inconsistent and incoherent national response” to the pandemic, “Americans must put a president in the White House come January, 2021, who will understand that public health should not be guided by partisan politics.”
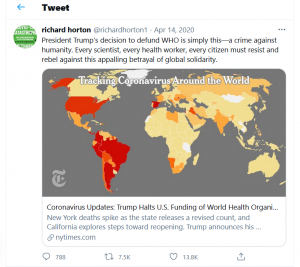
In April, right after President Trump announced the U.S. was pulling its funding of the WHO, Horton tweeted that Trump’s action was “a crime against humanity.”
Did Mr. Horton predict his own bias when he wrote in 2015: “In their quest for telling a compelling story, scientists too often sculpt data to fit their preferred theory of the world. Or they retrofit hypotheses to fit their data.”
Another revealing tidbit: the same week the fraudulent Mehra/Surgisphere study was submitted to The Lancet, editor Horton refused to publish Dr. Raoult’s study of 1,061 actual patients that showed positive results for HCQ.
Given the damage this fabricated study did in just a few weeks to legitimate research, imagine if Dr. Didier Raoult in France had not had the courage to attack its implausibility after just a few days paving the way for other scientists to question it as well.

This was also just a few weeks after he was unfairly attacked for his favorable reporting about the world’s most politicized drug. Notice the nearby photo of the cover of The New York Times Magazine published even before (10 days) The Lancet and NEJM Surgisphere studies were printed. Could they have made him appear more sinister?
BOULWARE STUDY
A much less publicized study but one that put another nail in the coffin of HCQ was a “gold standard” double-blind, RCT published in NEJM on June 2, 2020, just two days before the retraction of the Surgisphere fiasco. Though this study was not fraudulent, it is one considered “one of the most controversial studies in the history of medicine.”
It was led by Dr. David Boulware and dealt with the post-exposure prophylactic use of HCQ for COVID-19. The study focused on those who recently had close contact with someone who had tested positive for COVID. The actual individual in the study was not necessarily tested but all were given HCQ to determine its prophylaxis.
The authors concluded that though HCQ showed positive results, they weren’t statistically significant enough to warrant its use.
The Boulware study became the go-to study for all who wanted HCQ dead and buried. After all, it was a Fauci-favorite — a double-blind RCT. For many that meant it overruled all observational studies that had shown any results to the contrary. For the anti-HCQ crowd, this study is likely the one most pointed to as the definitive end to all pro-HCQ arguments.
But the biases of this study became legend.
Nothing in the Boulware study was mentioned about the participant selection process other than it was “random.” The raw data showed that subjects who took HCQ within two days of exposure were 38% less likely to develop symptoms. And given that a third of the subjects took the drug twice as long afterward or four days after exposure, the study was just marginally controlled. Additionally, just 75% of the (HCQ) subjects admitted to taking all of the medication.
Plus, “exposure” to a virus-infected person could have vary greatly among the participants. In this study, health care workers accounted for 2/3 of all subjects. Having “high exposure” was considered not having on personal protective equipment (PPE) for 10 minutes or more and less than 6 feet from a confirmed COVID-19 patient. (Being 5 feet away without an eye shield for 10 minutes was considered “moderate exposure” in the study. This is significantly different exposure from living 24/7 with a spouse or parent who has confirmed COVID-19.)
Of the 821 subjects who said they had high or moderate exposure, just 20 eventually tested positive for COVID-19. The Boulware authors combined laboratory tested subjects with those who had “illness compatible with COVID-19.” So they counted the “probables” and those who had “epidemiologic linkage” (high exposure) as COVID positives.
There is a legitimately “probable” category for COVID-19. But according to the CDC these are only between 8% and 12% of all confirmed cases. The Boulware study said these people had an illness with COVID-like symptoms but they accounted for a very high 79% of the 113 cases.
The Boulware authors said they were “unable to access diagnostic testing” during this time period. It is true that testing kits were in such demand last spring that many were hard to come by. It meant that many of the participants were never PCR tested so we don’t really know who had COVID and who did not.
The authors admitted HCQ proved safe. “There were no serious intervention-related adverse reactions or cardiac arrhythmias” to the use of HCQ, they concluded. Few in the media have cited that positive result.
Exhibiting virtually zero curiosity to drill down into the data, the anti-HCQ media got the results they were looking for. For them, HCQ, “Trump’s drug” was so dead and buried that even the president didn’t want to talk about it.
But it wasn’t over for many physicians and scientists. By this time they had seen too much anecdotal evidence and observational studies that showed HCQ was effective to just throw in the towel when so much was at stake.
Professor of statistics, Dr. Marcio Watanabe, at the Universidade Federal Fluminense in Brazil (one of the most prestigious universities in Latin America) reviewed the Boulware study data. He wrote that Boulware had actually misinterpreted his own data. Watanabe found statistical verification that the participants who got HCQ the earliest had the best outcomes. This was no surprise to many frontline physicians.
Dr. Watanabe found statistical evidence that HCQ had considerable prophylaxis when taken early. If taken the day of exposure, the “estimated relative reduction in symptomatic outcomes is 72%.” After one day the reduction is 49%; after two days it is 29%. After 3 days the results are, as Boulware said, statistically insignificant but this was quite a different conclusion from that of the original authors.
Dr. Watanabe sent his formal reinterpretation of the data to NEJM. It never responded.
SKIPPER STUDY
Three days after President Trump announced that HCQ could be a “game-changer,” Dr. Caleb Skipper began a double-blind RCT that sought to determine whether HCQ was safe and effective in non-hospitalized patients with COVID-19. It was published in early July in the Annals of Internal Medicine with an accompanying editorial that said this definitively ended the “sad saga” of HCQ.
The editorial’s conclusion can only be considered political, not scientific.
We already knew at the time that COVID-19 is clearly an old person’s disease. It has been from the beginning. Over the past year, nearly 500,000 Americans have died from the virus but the toll for those over 65 has been especially alarming. Though senior citizens (65+) make up less than 17% of the U.S. population, they’ve accounted for over 81% of the deaths.
The Skipper study admitted to having relatively young participants (77% were age 50 or under) and with few comorbid conditions. This is not the primary at-risk demographic (age 65+ with comorbidities). The study’s authors curiously stated that HCQ may actually work with older, at-risk patients: “It is possible that hydroxychloroquine is more effective in populations at higher risk for complications, such as older persons in long-term care facilities.“
This reported conclusion of this study was quite similar to the Boulware RCT. It must be stated that though these studies were RCTs, they were not “laboratory-monitored” but dependent on patients following instructions. The authors state this was “an internet-based trial.”
As such the patients were not even seen by the researchers to assess their symptoms; the patients simply filled out an emailed survey asking for a self-assessment. Additionally, it was a mail-in RCT as the patients were mailed their medications leaving questions about how early the drugs were first taken.
Contrast this to the Raoult study in which investigators physically saw each of their patients.
423 were enrolled in the Skipper study, 381 had laboratory confirmed COVID-19. Just 56% were enrolled within a day of disease confirmation, 43% some unstated time after that. 77% of the HCQ group admitted to completely following the dosage recommendations.
At day 14, the HCQ group had 20% “fewer ongoing symptoms” and 60% fewer hospitalizations.
Again, HCQ was used alone as the authors chose to ignore previous observational studies that highly recommended combining HCQ with azithromycin and zinc.
The authors concluded that “Change in symptom severity over 14 days did not differ between the hydroxychloroquine and placebo groups.”
But this was not true.
At day 14, the HCQ group had 20% “fewer ongoing symptoms” and 60% fewer hospitalizations. This at a time when all the talk was about “flattening the curve” which meant to keep hospitals and our medical system from being completely overrun.
This study, albeit with a small number of patients, should have concluded that this inexpensive, safe drug could keep COVID patients out of hospitals.
[A question comes to mind: If the authors found that there were significant benefits to giving HCQ early on and it prevented COVID patients from being hospitalized and perhaps dying, how would they ethically justify giving a placebo to the poor sacrificial “lab rats” who died? Wouldn’t concluding HCQ was effective have seemed ethically self-incriminating?]
This study was both flawed and produced an incorrect conclusion: “Hydroxychloroquine did not substantially reduce symptom severity in outpatients with early, mild COVID-19.” The key word here is “substantially.”
Of the 391 lab-confirmed COVID-19 cases, 201 got HCQ. 49 of them (24%) had symptoms after 14 days compared to 59 (of 194 who did not get HCQ) or 30% with placebo. Hospitalizations were cut in half with the HCQ group: 10 (5.2%) were hospitalized with placebo compared to 4 (2%) who received HCQ.
The Skipper study also resulted in an international “open letter” led by Dr. Marcio Watanabe and 42 medical statisticians and mathematicians regarding the misinterpretation of data compiled in these recent studies. While some physicians are well-versed in statistical analysis many are not.
The letter stated that “…Recent papers have had a substantial impact in the media, on public policies and within the scientific community. These three papers nevertheless share at least one common mistake: the conclusions they draw from their data are wrong.”
The letter’s signatories said that a correct conclusion would be to say that HCQ showed positive effects but that the results are statistically inconclusive and deserve further study. Or put another way, from these studies “it is not possible to affirm that early treatment of COVID-19 patients with hydroxychloroquine is not effective.”
A scientist in Dallas, Dr. David Wiseman, led a group including some from the Henry Ford Center of Medical Excellence that also reviewed the data from the Boulware and Skipper studies. They came to the same conclusion as Watanabe. Also reviewing the data were Alexander Chuan Yang, a professor at Wayne State University School of Medicine and Juan Luco in Argentina who said their conclusion was “fundamentally flawed.”
FORD STUDY
The Henry Ford Health System is a large, non-profit health care organization in Detroit. It performed a large-scale, peer-reviewed, highly controlled, observational study between March and May that yielded stunning results at the time. It found hospitalized patients who received HCQ for COVID-19 had a significant reduction in death rate.
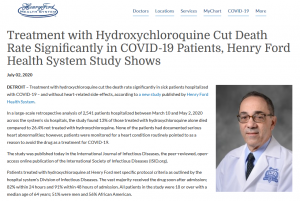
Over 2,500 COVID-19 patients who were hospitalized in the system’s six hospitals were retrospectively analyzed. The study was extremely detailed; it took into account 19 “confounding factors” and the severity of the illness in each of the participants.
The Ford system’s CEO of the Medical Group, Dr. Steven Kalkanis, released a statement stating, “Our analysis shows that using hydroxychloroquine helped save lives. As doctors and scientists, we look to the data for insight. And the data here is clear that there was benefit to using the drug as a treatment for sick, hospitalized patients.”
The death rate of those treated with HCQ alone was 13% compared to 26.4% of those who did not receive HCQ.
“Our analysis shows that using hydroxychloroquine helped save lives.”
Some questioned the veracity of the study given the different conclusions from the aforementioned studies. One of the Ford study’s authors, Dr. Marcus Zervos, attributed the discrepancies to close cardiac monitoring and “Our dosing also differed from other studies not showing a benefit of the drug. And other studies are either not peer reviewed, have limited numbers of patients, different patient populations or other differences from our patients.”
Dr. Zervos added, “What we think was important in our study … is that patients were treated early. For hydroxychloroquine to have a benefit, it needs to begin before the patients begin to suffer some of the severe immune reactions…”
The vast majority of the COVID-19 patients were administered HCQ early — 82% of them began treatment within 24 hours, 91% within 48 hours of admission.
Journalist Sharyl Attkisson related other details from the study: Fatality rate with neither HCQ or azithromycin – 26.4%; HCQ alone – 13%; Azithromycin alone – 22.4%; HCQ plus azithromycin – 21.1%.
In August, at the behest of its frontline physicians the Ford System requested from the FDA an exemption to continue its use of HCQ. The FDA refused.
On July 31st, when asked about the positive results from the Ford study and the use of HCQ on hospitalized patients, Dr. Anthony Fauci told a House subcommittee dismissively, “That study is a flawed study, and I think anyone who examines it carefully [will see] is that it is not a randomized placebo-controlled trial.”
A study of America’s news organizations revealed that by the end of July, U.S. media was thoroughly dug in against HCQ compared to the rest of the world. 50% of editorial reviews were negative compared to just 4.5% positive.
In August, at the behest of its frontline physicians the Ford System requested from the FDA an exemption to continue its use of HCQ.
The FDA refused.
META-STUDIES
If one controlled, objective study is good, two controlled, objective studies are better. In fact, the more the merrier. And if the studies all come down on the same side of the equation, the confidence level can legitimately go very high.
If the RCT is the “gold-standard,” the Meta-Study is the “platinum-standard.” It is the pinnacle of scientific analysis.
In view of all mentions of studies in this article, it is important to have an understanding of the “Meta-Study,” which combines smaller studies into one that has greater or complete “statistical significance.” This simply means the greater the number of observations, the lower the margin of error and the higher the degree of confidence in the accuracy of the results.
Here’s an example. We recently had a presidential election and heard endlessly about polling results. We listened to a pollster tell us something like: 2,144 likely voters were polled with the results showing that Candidate A has a 45% to 42% preference over Candidate B but well within the margin of error of 2%. (Had the number of voters polled been 1,014, the margin of error would be calculated to be 3%.)
This is a mathematical calculation in which the key inputs are the sample size and confidence level sought. The above example uses a 95% confidence level which is fairly typical. See the calculator here. Another way to say it is Candidate A has a slight lead over B but it is not statistically significant.
Now using the same example, on the same day another pollster finds candidate A with the same 45% to 42% preference over B and this pollster asks the same number of likely voters. Again, the margin of error is 2% with a 95% confidence level.
Individually, neither poll gives Candidate A a statistically significant lead BUT —and this is where the “Meta-Studies” come in— taken together, Candidate A indeed has a statistically significant lead or an actual lead in the race. Together the two polls got results from 4,288 voters which when calculated gives a margin of error of just 1.33%. Adding 1.33% to Candidate B’s 42% and taking 1.33% away from A’s still leaves Candidate with the lead. A’s lead is now outside the margin of error.
This is the same principle used in medical research. It’s how Meta-Studies can mathematically alter the actual meaning of individual studies. It’s where doctors may step aside and mathematical statisticians enter the picture. A number of seemingly inconclusive smaller studies can have a large consequential impact when taken together.
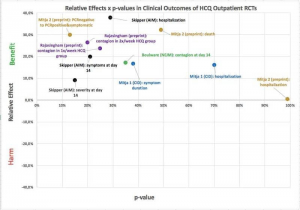
Mathematician Flavio Abdenur conducted a meta-analysis of all the “gold-standard” RCT studies involving HCQ and placebos. He put it in graphic form and published it on his Facebook page in October (Figure X). He also made available all data involved in his calculations.
Each of these studies found some benefit to administering HCQ but each author also reported that his or her findings were not statistically significant mostly because the studies were relatively small — just a few hundred patients. In other words they were statistically under-powered.
“Conclusion– Hydroxychloroquine use in outpatients reduces the incidence of the composite outcome of COVID-19 infection, hospitalization, and death.”
In the Flavio chart notice that all of the outpatient RCTs examining HCQ showed positive outcomes. This meta-analysis showed conclusively that HCQ is statistically effective as an outpatient treatment.
Yale epidemiologist, Dr. Harvey Risch, performed a meta-analysis that went beyond what Drs. Watanabe and Flavio Abdenur calculated.
Dr. Risch’s meta-study included 5 RCTs that enrolled 5,577 patients. He and the other authors found that “HCQ was associated with a 24% reduction in COVID-19 infection, hospitalization or death… Conclusion– Hydroxychloroquine use in outpatients reduces the incidence of the composite outcome of COVID-19 infection, hospitalization, and death.”
This was posted on September 30, 2020 and remains the true state of the science regarding HCQ for early treatment.
The granddaddy of all meta-studies is provided here. It is an ongoing, up-to-date comprehensive site that includes every study published world-wide that deals with HCQ and COVID-19.
So far there have been 26 total studies to determine if HCQ reduces the need for hospitalization and/or reduces mortality. Of these 26 studies, 100% have produced positive results with a 66% average improvement.
This summary shows so far there have been 26 total studies to determine if HCQ reduces the need for hospitalization and/or reduces mortality. Of these 26 studies, 100% have produced positive results with a 66% average improvement.
It’s interesting to note that there have been 137 total studies looking at HCQ’s efficacy in late treatment. Even here, HCQ had positive outcomes in 102 or 74.5% of these.
None of these meta-studies have been published in one of the primary medical journals. Why not? Well, since no one has challenged the calculations or the validity of these meta-study conclusions, the lack of interest by the entrenched medical-industrial complex can only be due to politics, not science. In fact, anyone who ignores or disparages the results of these meta-studies that show the efficacy of HCQ is in fact a denier of science.
BIAS
In 2005, Dr. John Iaonnides published a paper in the Journal of Public Library of Science (PLOS), that became the most downloaded in the history of the publication entitled, “Why Most Published Research Findings Are False.” Dr. Iaonnides said all studies have design flaws and biases. Some are unintended, some not.
A few years ago an article in the journal Nature said that there was a “replication crisis” with medical studies. The astonishing article said, “More than 70% of researchers have tried and failed to reproduce another scientist’s experiments, and more than half have failed to reproduce their own experiments.” This revelation did not surprise many frontline physicians.
“More than 70% of researchers have tried and failed to reproduce another scientist’s experiments, and more than half have failed to reproduce their own experiments.”
In 2014 The Journal of American Medical Association (JAMA) published a study, again by the Ioannidis group, that found 35% of the most respected RCTs could not be replicated using the studies’ own raw data. This is not comforting news to lay people like myself.
Another study by the NIH looked at 52 RCTs and found that many authors (researchers) in their zeal to eliminate the confounding factors in their study often excluded many patients who were far more representative of the general population.
RCT fundamentalists are those who ignore all other available information like observational studies and only focus on RCT strengths, never on their weaknesses.
For example, the NIH published an article that examined 15 oncological RCTs in which the authors found that “real-world patients with cancer were often older, and more likely to be female, have a poor performance status, and worse disease prognosis.”
This is why medical curricula, major journals and texts publish not just RCTs but observational studies, case histories and others for frontline physicians to examine the weight of the evidence in caring for their patients.
MEDICAL – INDUSTRIAL COMPLEX
Federal Bureaucracy
Recently Dr. Anthony Fauci, head of the NIAID but the face of the federal healthcare bureaucracy, signaled that he finally feels free to speak honestly about Covid-19 now that former President Donald Trump is out of office.
Dr. Fauci said that the new administration was committed to being “completely transparent, open and honest,” a sharp break from the Trump White House, when Fauci said he often felt there would be repercussions for speaking honestly about the pandemic.
“It was very clear that there were things said, be it regarding things like hydroxychloroquine and other things like that, that really was uncomfortable because they were not based in scientific fact.” Dr. Fauci said he had just met with Biden and discussed how “everything we do will be based on science and evidence.”
So far, that has not been the case.
Dr. Anthony Fauci has been a Washington bureaucrat for decades. He hasn’t always been so adored by the media. Over 30 years ago, the country was facing thousands of deaths due to HIV/AIDS. At the time, Dr. Fauci was the Federal Czar on AIDS. At a tense meeting in 1987, AIDS activists Michael Callen and Dr. Barry Gingell were literally begging Dr. Fauci to formally permit use of the drug, Bactrim, as a prophylaxis for AIDS-induced pneumonia. This was being successfully used by many frontline doctors treating the disease.
Fauci refused. He said there was no “gold standard” RCT showing Bactrim’s efficacy. The liberal Huffington Post was especially critical of Dr. Fauci. It said, “Anthony Fauci is rewriting history. He is doing so to disguise his shameful role in delaying promotion of an AIDS treatment that would have prevented tens of thousands of deaths in the first years of the epidemic.”
At the time, his demand for an RCT was an unusual position for Dr. Fauci. Some years before he had pushed for the approval of a treatment for Wegener’s Granulomatosis as the author of an anecdotal study with only 18 patients using no controls and no placebo.
But Dr. Fauci had no problem with the federal green light for Burroughs Wellcome’s lucrative drug, AZT, to use against HIV/AIDS which was approved in record time. AZT wasn’t all that effective, had strong side effects and cost about $8,000/year per patient.
Time magazine wrote, “BW conducted a (small sample) trial in which the group receiving AZT had just one death and the controlled group had 19 deaths. So they stopped the trial after 16 weeks as it “appeared” the drug was effective. AZT was fast-tracked by the FDA and approved in just 20 months.”
And finally, after thousands of lives had been lost, Bactrim was formally approved in 1989.
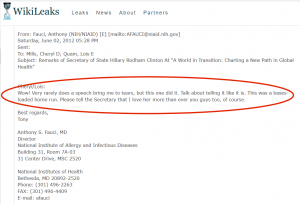
Dr. Fauci has been portrayed by the media as being a pure man of science and above the political fray. But he was clearly a major Hillary Clinton fan a few years ago. He said he heard a speech of hers back in 2012 and was moved “to tears.” Would he have given HCQ, “Trump’s drug,” a fair shake? Maybe. Maybe not.
Israel’s Tel Aviv University’s prestigious Dan David Prize of $1 million was just awarded to Dr. Fauci as “the consummate model of leadership and impact in public health.”
Gilead and Remdesivir
Gilead is a pharmaceutical powerhouse with an annual revenue of over $22 billion. Its COVID entry, (brand name: Veklury) remdesivir, was originally developed for Ebola. It had only marginal success and was reissued to treat COVID-19. Gilead saw a potential gold mine.
In the first quarter of 2020, Gilead dramatically ramped up its lobbying efforts in Washington by spending 32% more than it had in Q1 2019. The bureaucratic “marketing” effort was deemed worth it to Gilead. Remdesivir is very expensive to purchase. It costs up to $3,100 per patient for a COVID-19 treatment.
Given this golden opportunity, Gilead was quite willing to fund a “gold standard” trial for its prized drug. But the trial took a bit of a turn mid-stream when, as The Washington Post reported, “Government clinical trial investigators changed the primary metric for measuring the success of Gilead’s experimental drug remdesivir as a coronavirus treatment two weeks before Anthony S. Fauci’s announcement that the drug would be the new ‘standard of care.’”
Since the study showed the drug did not reduce mortality, the study had to be reworked to show something positive. Use of the drug apparently reduced the recovery time of those who lived from 15 days to 10 so this became the centerpiece.
Dr. Fauci made an optimistic announcement about the study in the Oval Office on April 29th. Within 2 months, Gilead had signed a $1.2 billion agreement with the United States government to provide 500,000 treatments of remdesivir.
While Gilead was becoming the darling of the D.C. medical bureaucracy, it was still paying a $97 million fine levied against it by the U.S. Department of Justice for paying kickbacks under the False Claims Act.
Remdesivir is not without its risks. After Gilead’s agreement with the U.S., a later study said that of five hospitalized patients who were being treated with remdesivir, four of the five had to have their treatment discontinued due to “ALT elevation and/or renal failure.” There has been very little subsequent media scrutiny regarding this red flag.
In August, The Washington Times reported that The Lancet study released in May was repudiated “in large part (due) to an ophthalmologist named, Dr. (James) Todaro.”
“When the president mentioned hydroxychloroquine, Gilead’s stock fell 8.7% within hours and continue to fall into the following week erasing $21 billion from Gilead’s market cap.”
At a highly publicized presentation by frontline doctors in Washington last July, Dr. Todaro spoke about why Gilead would have considered HCQ a serious economic threat. He said the public had a misconception that there was no coordinated attack on HCQ.
Dr. Todaro said, “Minutes before the president gave his press conference on March 19th where he first mentioned hydroxychloroquine at 11:31 in the morning, Gilead’s stock was trading at $85 per share so this was higher than it had been in the previous two years. When the president mentioned hydroxychloroquine, Gilead’s stock fell 8.7% within hours and continue to fall into the following week erasing $21 billion from Gilead’s market cap.”
For Gilead’s stock to recover required positive remarks about remdesivir by Dr. Fauci at a press conference about six weeks later.
Dr. Todaro said, never mind that remdesivir was a “failed hepatitis C drug with a fairly toxic profile and with side effects (but has been given) almost a complete pass.”
Shortly after singing remdesivir’s virtues, Dr. Fauci was quick to condemn HCQ following the publication of the “Lancet” study. Dr. Todaro said that Dr. Fauci went on CNN and reinforced the false notion that HCQ causes cardiovascular problems.
Regarding that retracted “Lancet” study, Dr. Todaro said he could not speak for the motivations of the authors but the two lead physicians have ties to Gilead and have spoken highly of remdesivir. Dr. Todaro stated, “Dr. (Mandeep) Mehra, the study’s lead author, was also one of the lead speakers on a very select list of speakers for a conference in April on COVID-19 that was sponsored by Gilead.”
The damage done to HCQ from the previously cited studies must be compared to Gilead’s march toward FDA approval for remdesivir. In April, the FDA had given EUA to Gilead based on very modest results in preliminary tests for remdesivir. Mortality in just over 1,000 COVID patients was reduced to 8% for those on remdesivir from 11.6% in the control group. Positive? Yes, but as Skipper might say, not statistically significant.
The final and largest study, the WHO’s “Solidarity” trial, found that the drug (remdesivir) did nothing to reduce time of recovery nor mortality.
The EUA though continued into the fall. In early October, Gilead signed a lucrative agreement with the European Union to purchase worth more than $1 billion of the drug.
This caught many scientists by surprise since two of the most recent (relatively small) studies had shown no benefit at all. And the final and largest study, the WHO’s “Solidarity” trial, found that the drug did nothing to reduce time of recovery nor mortality.
The New York Times thought remdesivir was finished as a possible therapy for COVID.
The WHO study was released on October 15th. That same day, Gilead quickly released a statement trying to mitigate the damage.
[The study by the WHO was slightly surprising given that Gilead is a substantial contributor to the World Health Organization. In fact these countries actually gave less to the WHO in the current 2020-21 fiscal year than did Gilead: New Zealand, Austria, Indonesia, Finland, Egypt, Singapore, Greece, Portugal and Thailand. The largest single contributor ahead of even the U.S and Germany? The Bill and Melinda Gates Foundation gave nearly 12% of the organization’s entire budget.]
Gilead’s response must have succeeded because just one week later, on October 22nd, a shocking announcement came from the FDA saying it was formally approving the use of remdesivir for treating hospitalized patients with COVID-19. FDA Commissioner Stephen M. Hahn, M.D. released a statement: “Today’s approval is supported by data from multiple clinical trials that the agency has rigorously assessed and represents an important scientific milestone in the COVID-19 pandemic.”
Science magazine’s headline right after the FDA approval of remdesivir said, “The ‘very, very bad look’ of remdesivir, the first FDA-approved COVID-19 drug.”
As the American Association for the Advancement of Science (AAAS) reported, the FDA’s decision “baffled scientists.” AAAS said the FDA never consulted a group of outside experts that it has at its disposal just for such consultations. This group is called, the Antimicrobial Drugs Advisory Committee (AMDAC) and includes infectious disease clinicians, biostatisticians and pharmacists. Not only was the group not consulted for remdesivir but according to AAAs, “it has not convened once during the pandemic.”
It seems reasonable to ask, why not?
* * *
The U.S. pharmaceutical industry is enormous. It accounts for over $1 trillion in economic activity or about 1/25 of the country’s entire economic output. In the history of capitalism there’s likely never been an industry that grew in size and strength that at some point, didn’t try to protect itself from perceived threats. This industry is no different.
“[The pharmaceutical industry is] now primarily a marketing machine. … this industry uses its wealth and power to co-opt every institution that might stand in its way, including the US Congress, the FDA, academic medical centers, and the medical profession itself.”
The aforementioned Lancet editor, Richard Horton, is certainly a political animal but he can also be refreshingly candid. In 2015 he wrote, “The case against science is straightforward: much of the scientific literature, perhaps half, may simply be untrue. Afflicted by studies with small sample sizes, tiny effects, invalid exploratory analyses, and flagrant conflicts of interest … science has taken a turn towards darkness.”
Marcia Angell, former editor of the NEJM, agrees with Horton. She wrote, “[The pharmaceutical industry is] now primarily a marketing machine to sell drugs of dubious benefit, this industry uses its wealth and power to co-opt every institution that might stand in its way, including the US Congress, the FDA, academic medical centers, and the medical profession itself.”
The BMJ (formerly the British Medical Journal) is one of the world’s most important medical publications. In an editorial entitled, “COVID-19: politicization, corruption and suppression of science,” editor Kamran Abbasi wrote, “Science is being suppressed for political and financial gain. Covid-19 has unleashed state corruption on a grand scale, and it is harmful to public health. … The medical-political complex tends towards suppression of science to aggrandize and enrich those in power. And, as the powerful become more successful, richer, and further intoxicated with power, the inconvenient truths of science are suppressed. When good science is suppressed, people die.”
“And, as the powerful become more successful, richer, and further intoxicated with power, the inconvenient truths of science are suppressed. When good science is suppressed, people die.”
Physicians and Professional Organizations
In early November the Journal of the American Medical Association (JAMA), the most highly regarded of all medical journals, published an op-ed entitled, “Misguided Use of Hydroxychloroquine for COVID-19.”
The author, Dr. Michael Saag, mentioned nothing about early treatments using HCQ and the dozens of studies published by last fall showing HCQ’s proven efficacy. So the question becomes, Was this omission intentional?
It’s impossible to say but Dr. Saag did report that his institution had received grants from Gilead.
Both the NEJM and The Lancet have put out similar position statements. In a scathing article from 2005, Richard Smith, long time editor of the BMJ wrote, “Medical Journals Are an Extension of the Marketing Arm of Pharmaceutical Companies.”
Smith gave us a stunning look into the “above reproach” medical-industrial complex. He wrote, “Overall, studies funded by a company were four times more likely to have results favorable to the company than studies funded from other sources.” (My emphasis)
The BMJ editor added, “The evidence is strong that companies are getting the results they want, and this is especially worrisome because between two-thirds and three-quarters of the trials published in the major journals — Annals of Internal Medicine, JAMA, Lancet, and New England Journal of Medicine — are funded by the industry.” (My emphasis)
“Journals have devolved into information laundering operations for the pharmaceutical industry.”
The publishers of these journals are well aware that drug companies buy tons of advertising and thousands of dollars worth of reprints. As the editors are more and more responsible for the budgets of their publications, they are increasingly compromised by what they might consider economic necessities.
Smith quoted Richard Horton, chief editor of The Lancet, “Journals have devolved into information laundering operations for the pharmaceutical industry.”
* * *
Ignoring now dozens of studies showing both the safety and effectiveness of the HCQ cocktail for COVID-19, the American Medical Association (AMA) remains adamant in its refusal to sanction HCQ use. A resolution was placed on its November 2020 meeting agenda by a frustrated group of frontline physicians seeking to secure formal AMA support for the ability to “prescribe an FDA-approved medication for off-label use, if it is in her/his best clinical judgement, with specific reference to the use of hydroxychloroquine and combination therapies for the treatment of the earliest stage of COVID-19.”
The resolution failed.
But at that same special November meeting the AMA did take some action. Its website’s headline reads, “Top 10 stories from the November 2020 AMA Special Meeting.” One might think the majority of the ten action items might deal with the worst pandemic in a century but — just one did: #3 – Spelling out physicians’ duties on COVID-19 vaccination.”
Among the other critically important issues that the AMA wanted to address at this “special” meeting were:
• Physicians back new approaches to cover more of the uninsured.
• Racism is a threat to public health.
• AMA to help shape clear criteria for compassionate release of inmates.
• Workplace bullying must have absolutely no place in medicine.
• Law enforcement’s excessive use of force is a public health issue.
• When patients are prejudiced, here’s what physicians should do.
On its website, that apparently hasn’t been updated since August, the AMA gives official guidance to its member physicians. One of the questions of our time is: How do I treat patients with confirmed COVID-19? The AMA responds:
- “People with COVID-19 should receive supportive care to help relieve symptoms either at home or in a clinical setting as symptoms demand. For severe cases, treatment should include care to support vital organ functions.
- “At present, no drug has been proven to be safe and effective for treating COVID-19. There are no Food and Drug Administration (FDA)-approved drugs specifically to treat patients with COVID-19.”
- “Some patients with COVID-19 have received intravenous remdesivir, an investigational antiviral drug that was reported to have in-vitro activity against SARS-CoV-2, for compassionate use outside of a clinical trial setting.
- “As of June 15th, the FDA has revoked emergency use authorization of hydroxychloroquine and chloroquine to treat COVID-19.
- “In the event that an adverse drug event is suspected or observed from any medication used to prevent or treat COVID-19, we urge health care providers to submit a report to FDA MedWatch. COVID-19 or coronavirus should be referenced in the report to the FDA.
- “Read the NIH Coronavirus Disease 2019 (COVID-19) Treatment Guidelines.”
So the primary physician organization in the United States, after a year into the pandemic has zero medical-related recommendations for early treatment. Comforting.
And its statement, “At present, no drug has been proven to be safe and effective for treating COVID-19” is factually untrue.
On the other hand, another primary physicians’ organization representing all specialties since 1943, The Association of American Physicians and Surgeons (AAPS) has been greatly focused on treating patients with COVID-19, including early treatment not just those hospitalized.
Some of its headlines over the last 7 months or so posted on its website include:
- Posted: July 7, 2020. New Study Shows That Hydroxychloroquine Saves Lives, States AAPS.
- Posted: July 22, 2020. More Evidence Presented for Why Hydroxychloroquine Should be Made Available, in a New Court Filing by AAPS.
- Posted: August 26, 2020. Support HJ 5002 and Access to Hydroxychloroquine for COVID-19.
- Posted: August 31, 2020. COVID-19: Might Hydroxychloroquine Actually Be Good for Your Heart?
- Posted: October 13, 2020. FDA Can Provide No Justification for Warnings on Hydroxychloroquine.
- Posted: December 17, 2020. AAPS Sues the FDA to End Its Arbitrary Restrictions on Hydroxychloroquine.
- Notice any differences with the focus of the two professional organizations?
CONCLUSION
There is little in our world today that hasn’t been “politicized” i.e., manipulated to fit a political or ideological narrative. One might think that something as seemingly “objective” as science could remain unscathed but it has become one of the most politically — and economically — manipulated.
It has been routine for the news media to invoke “science” whenever it published anything negative regarding hydroxychloroquine. Last spring the media really didn’t know if HCQ worked or not, but just like Fox News Channel’s Neil Cavuto shouting to his viewers, “This will kill you,” since it was “Trump’s drug,” they really didn’t care.
The national media were ideologically predisposed to believe that any cheap, supposedly safe drug promoted Donald Trump just couldn’t work. Or, and a far more sinister possibility is they thought it might be effective — but didn’t want it to be. They knew its success would likely guarantee Trump’s reelection and that possibility was too appalling to contemplate.
I’m neither naive enough to think that there weren’t political operatives who, as Rahm Emanuel famously urged not waste a good crisis nor cynical enough to think the pandemic and its devastating continuation was intentional.
The media pointed out the negative studies — as they should have. But they did, and continue to censor the many more positive studies that have shown how using HCQ early in the illness saves lives.
In just two months after Trump’s “game changer” announcement in March, a Google search found 160 reports in which hydroxychloroquine was characterized as “unproven” regarding COVID-19.
None mentioned that many useful, life-saving drugs in the past have only been studied “observationally.” Nor that statistical analysis can show that a number of observational studies taken together can produce far more important and accurate information than an RCT. Instead, like Dr. Fauci, they trashed them.
Last spring a great divide also developed between the researcher/scientist and frontline physician. Doctors who were actually seeing frightened, high-risk patients in the flesh knew they were in a viral war-zone with little or no federal guidance.
After Drs. Raoult’s and Zelenko’s studies were published last spring, many physicians began to prescribe HCQ (along with zinc and azithromycin) for their COVID-19 patients. They were similarly successful.
More observational” studies began to show up everywhere. These were (still are) ignored by bureaucrats along with many professional organizations like the American Medical Association (AMA).
Doctors were finding that their patients who took the HCQ cocktail were doing much better than thousands of COVID-19 patients whose physicians were following CDC and NIH guidelines and prescribing — nothing. To continue to assert, like Dr. Fauci, that all these successes are simply anecdotal coincidences is woefully unscientific.
Now one full year after the beginning of this destructive pandemic, this is the official guidance from the Centers for Disease Control:
“Take care of yourself. Get rest and stay hydrated. Take over-the-counter medicines, such as acetaminophen, to help you feel better. Stay in touch with your doctor. Call before you get medical care. Be sure to get care if you have trouble breathing, or have any other emergency warning signs, or if you think it is an emergency.”
Yale epidemiologist, Dr. Harvey Risch, also testified that “our government research institutions have invested billions of dollars in expensive patent medication and vaccine development but almost nothing in early outpatient treatment, the first line of response to managing the pandemic.”
“Every study of outpatient use of one drug, hydroxychloroquine, with or without accompanying agents, has shown substantial benefit in reducing risks of hospitalization and mortality.”
He said, “What I have observed is that … every study of outpatient use of one drug, hydroxychloroquine, with or without accompanying agents, has shown substantial benefit in reducing risks of hospitalization and mortality.”
According to Dr. Lee Merritt, orthopedic spine surgeon, there are just five states in the United States where HCQ can be prescribed for COVID-19. Though it may be less toxic than Tylenol, a physician is not permitted to independently and expertly view all the evidence about an FDA-approved drug and decide for him or herself what is the best course of action for the patient.
The FDA’s (including governors and pharmaceutical boards) ban of HCQ as an off-label prescription is unprecedented. As many as 20% of all prescriptions or about 100 million are given for off-label use. Politically removing this proven safe therapy from the physician’s arsenal against the deadly virus is morally unconscionable.
HCQ has been prescribed hundreds of millions of times safely for over 65 years. It has been taken by tens of millions of people. It is sold over-the-counter in many parts of the world. The CDC says it is safe for everyone including children and pregnant women. Some states have such an excess supply of HCQ they don’t know what to do with it. Yet if the physicians try to prescribe it, they risk losing their medical license.
Hydroxychloroquine for early treatment is no longer a scientific uncertainty. As of the date of this publication, 26 studies have analyzed the results of 18,751 patients. Each study has shown a positive outcome; a 66% average improvement in the patient’s condition. (See appendix)
This drug is cheap, safe, widely available and proven it works. Why is it too much to ask to allow doctors digest all the available information and decide for themselves what’s best for their patients?
* * *
APPENDIX:
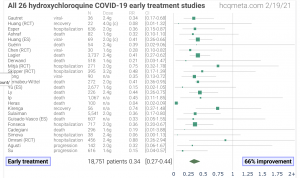
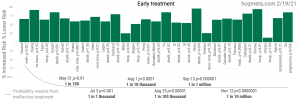
Public Comments from Frontline Physicians
- “We actually had a treatment for this that works extremely well in spite of all the propaganda and the attempts to falsify the medical literature which they’ve got caught at and the attempts to dismiss anything they don’t agree with.”
- “So you say why would they hide treatment? Well, I can come up with two reasons one is that your $69 billion vaccine industry goes to 0 if you have an effective treatment for all these viral airborne diseases.”
- “I would just make this point to doctors. I get it if you’re in training and you can’t speak out you’re stuck you’re not I don’t fault you. I do fault everybody above that the people that are taking the money from Fauci; the people that are taking the money from the NIH that are willing to take that money and push remdesivir. They’re killing them by omitting treatment, early honest outpatient (treatment) that works.”
- “We (doctors) need to man up and be honest here. Don’t tell me there’s no evidence you know they’re lying to you about the evidence if you really make any effort on the Internet you can find the evidence.
- “This is the first time historically that we’ve told patient with a disease to go home and isolate. …We’re letting patients perish unnecessarily. “Doing nothing is completely against the hippocratic oath.”
Dr. Mark McDonald (Child and adolescent psychiatrist):
- “I can tell you that the state of our children is abysmal. I have seen a substantial and rapid growth in the emotional illness of all of the children in my practice. Every single one.”
- “Anxiety, depression, suicidal ideation, self harm, child abuse, violence, panic attacks. I could go on and on.”
- “The pandemic in my mind is not so much of a medical pandemic as it is an emotional pandemic… It is based on, and centered around fear.”
- “This was a drug that was not controversial; is actually sold in the vitamin section of the stores across the world.”
- “The only two things you need to know about a drug is: Is it safe and is it effective? It almost doesn’t matter if something works we well. Lots and lots of things don’t work right. That’s capitalism. You can buy whatever you like. Here the question is: is it safe? The government’s obligation to make sure things that are being sold [are] safe.”
Dr. Scott Barbour: (orthopedic surgeon)
- “This VA study took me less than 60 seconds to read it and realize it is retrospective on older vets with tremendous comorbidities no zinc and used at toxic levels. Of course that wasn’t going to work and I think there was an attempt to sort of discredits hydroxychloroquine.”
- “For anybody’s who’s published a research paper and I have, it is tough. It takes a long time. You have to get copies of it together, it has to get peer reviewed, it gets sent back and forth, there are all kinds of revisions and things that have to be done.”
- “The peer reviewers at the New England Journal of Medicine and The Lancet — I can’t accept that it was just a random mistake. Something crazy is going on here.”
- “So the Lancet in the New England Journal of Medicine they retracted these studies but what happened as a result – the first thing was we ended the emergency use authorization for hydroxychloroquine. Governors around the country made it illegal to prescribe hydroxychloroquine, this safe medication again that’s been around 65-70 years.”
- “Once a drug is FDA approved, as a physician I am allowed to use that drug provided I use the dose that’s recommended for the FDA approved interval and duration that it’s approved for and if I have the consent of the patient that’s all I need to prescribe this drug.”
- “They threatened pharmacists so that if a doctor wrote the prescription the pharmacist would refuse to fill the medication and thereby effectively preventing its use.”
Dr. James Todaro: (Ophthalmologist)
- “The NIH COVID-19 treatment panel is made up of about 50 experts that are the ones that may be decisions on … what doctors should and should not use for COVID-19 and about … 9 of the experts sitting on the panel are either employees of Gilead have received financial support from Gilead. So almost 20% of that panel has a conflict of interest with Gilead.”
- “A lot of academic medical centers receive large grants from these pharmaceutical companies and then they receive grants from the NIH and other branches of government to conduct studies on these pharmaceuticals and so they may be inclined not to harm those relationships that (jeopardizes) ….those grants in the future.”


Good article, but it misses a significant point: HCQ is most effective as pre-exposure prophylaxis (PrEP). There are now five cohort studies from India that support this observation. Unfortunately HCQ PrEP threatened to slaughter with the vaccine sacred cow, so that application had to be crushed at any cost to allow the experimental dissemination of COVID vaccines. Maybe Chapter Two of this sad political story. See links:
https://www.webwire.com/ViewPressRel.asp?aId=265075
https://www.24-7pressrelease.com/press-release/478930/covid-19-linked-to-devastating-pregnancy-complications